Key Takeaway
Purified clinoptilolite-tuff significantly improved stool consistency and reduced days with loose stools in IBS-D patients compared to placebo in a 12-week RCT.
Summary
This randomized, double-blind, placebo-controlled trial investigated the efficacy and safety of purified clinoptilolite-tuff (zeolite) in 41 patients with diarrhea-predominant irritable bowel syndrome (IBS-D). Participants received either 3 g/day of clinoptilolite or placebo for 12 weeks, with a 4-week follow-up period.
The zeolite group showed statistically significant improvements in stool consistency as measured by the Bristol Stool Scale, with a meaningful reduction in the number of days per week with loose or watery stools compared to placebo. The treatment was well-tolerated with no significant adverse effects reported, confirming the favorable safety profile of purified clinoptilolite.
This is one of the few rigorous RCTs specifically examining zeolite for IBS-D, and the positive results provide moderate-quality evidence supporting clinoptilolite as a safe, non-pharmacological option for managing diarrhea-predominant IBS symptoms. The mechanism is thought to involve zeolite's ion-exchange and adsorptive properties in the gut lumen, helping to normalize intestinal fluid balance.
Figures
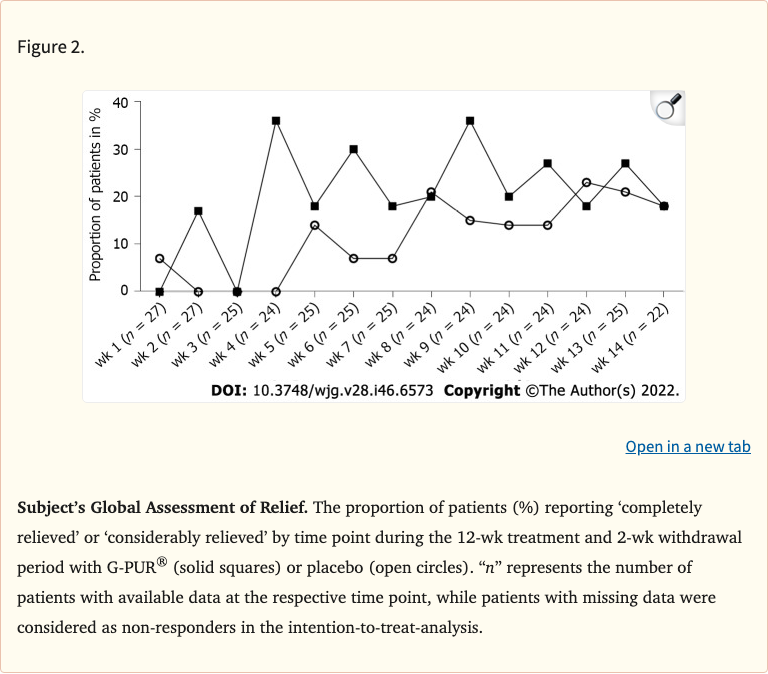 Figure 1
Figure 1
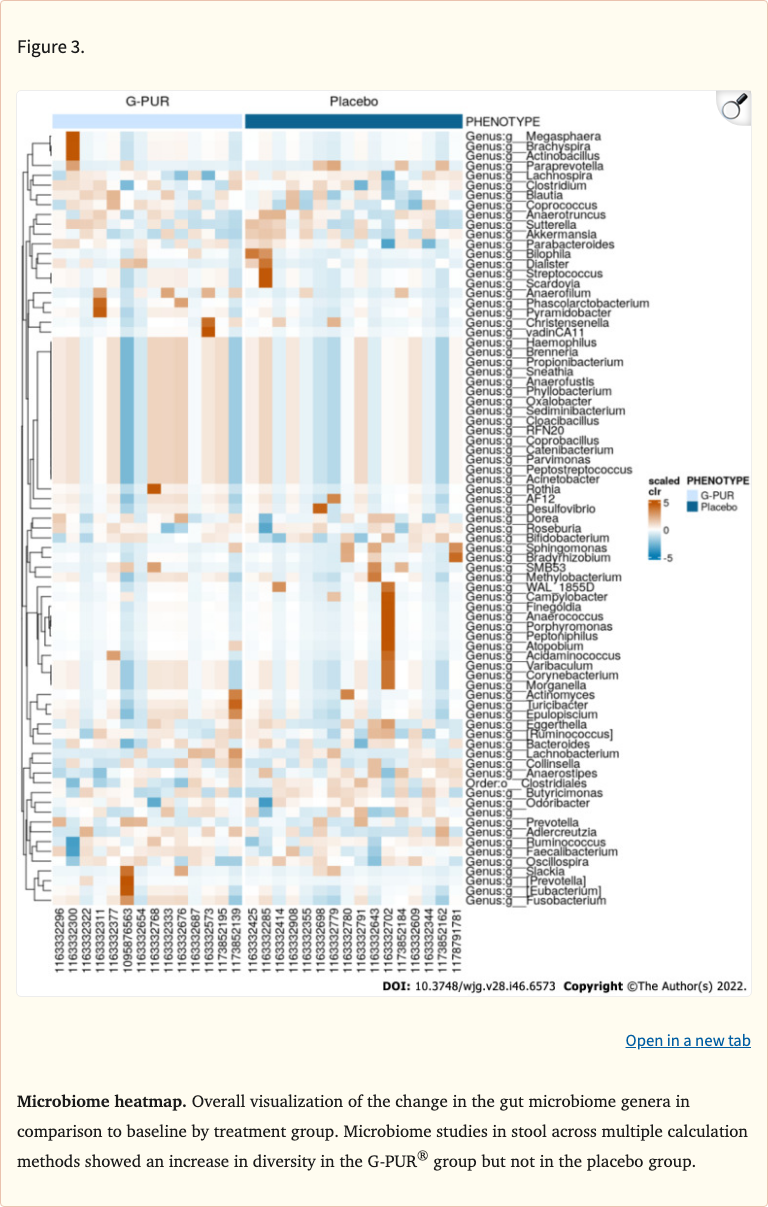 Figure 2
Figure 2
 Figure 3
Figure 3
 Figure 4
Figure 4
