Key Takeaway
Clinoptilolite-zeolite supplementation improved stool consistency and reduced IBS symptoms across all subtypes in a real-world observational study of 82 patients.
Summary
This non-interventional (observational) study evaluated the use of PMA-zeolite (panaceo med acute, a purified clinoptilolite product) in 82 patients diagnosed with irritable bowel syndrome (IBS) across all subtypes — IBS-D (diarrhea-predominant), IBS-C (constipation-predominant), and IBS-M (mixed). Patients took zeolite for 12 weeks and were assessed using the Bristol Stool Scale and IBS-SSS (Symptom Severity Score).
Across all three IBS subtypes, stool consistency improved significantly toward the normal range on the Bristol Stool Scale. IBS-D patients saw their scores decrease (firmer stools), while IBS-C patients saw scores increase (softer stools), and IBS-M patients stabilized. Overall symptom severity as measured by IBS-SSS also improved substantially, with the mean score dropping from moderate-severe to mild categories.
As a non-interventional study without a placebo control, the evidence is limited by potential placebo effects and reporting bias. However, the results are consistent with findings from controlled trials of clinoptilolite in IBS and suggest broad applicability across IBS subtypes, supporting its role as a complementary gut health intervention.
Figures
 Figure 1
Figure 1
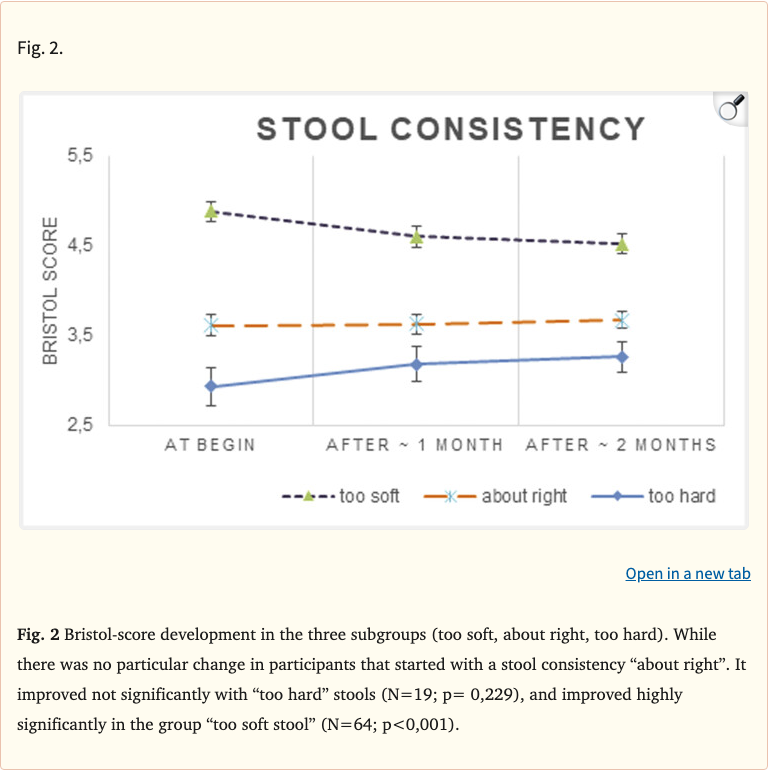 Figure 2
Figure 2