Key Takeaway
The largest TENS meta-analysis ever conducted (381 studies) found TENS significantly reduces pain intensity compared to placebo, with effects observed across both acute and chronic pain conditions.
Summary
This landmark meta-analysis - the largest ever conducted on TENS - systematically reviewed 381 studies to definitively assess the efficacy and safety of TENS for pain management in adults. The "meta-TENS study" addressed longstanding questions about TENS effectiveness that smaller reviews could not conclusively answer.
The researchers analyzed data across a wide range of acute and chronic pain conditions, providing the most comprehensive evidence synthesis to date. The sheer volume of included studies allowed for robust subgroup analyses by pain type, TENS parameters, and comparison conditions.
The results confirmed that TENS produces statistically significant and clinically meaningful pain relief compared to placebo/sham TENS across both acute and chronic pain populations. The safety profile was excellent, with minimal adverse events reported. This study represents the definitive evidence base supporting TENS as an effective non-pharmacological pain management tool.
Methods
- Comprehensive systematic review following PRISMA guidelines
- 381 studies included from extensive database searches
- Included randomized controlled trials comparing TENS to placebo/sham or no treatment
- Separate analyses for acute pain and chronic pain conditions
- Primary outcome: pain intensity (various scales standardized)
- Secondary outcomes: analgesic consumption, adverse events, function
- Random-effects meta-analysis with standardized mean differences
- Extensive subgroup and sensitivity analyses
- GRADE assessment for certainty of evidence
Key Results
- TENS significantly reduced pain intensity vs placebo/sham (SMD favorable for TENS)
- Effects significant for both acute and chronic pain conditions
- Pain relief observed across multiple pain etiologies
- Reduced analgesic consumption in some analyses
- Very low incidence of adverse events (mostly mild skin irritation)
- No serious adverse events attributable to TENS
- Effects maintained across sensitivity analyses
- Moderate certainty evidence overall
Figures
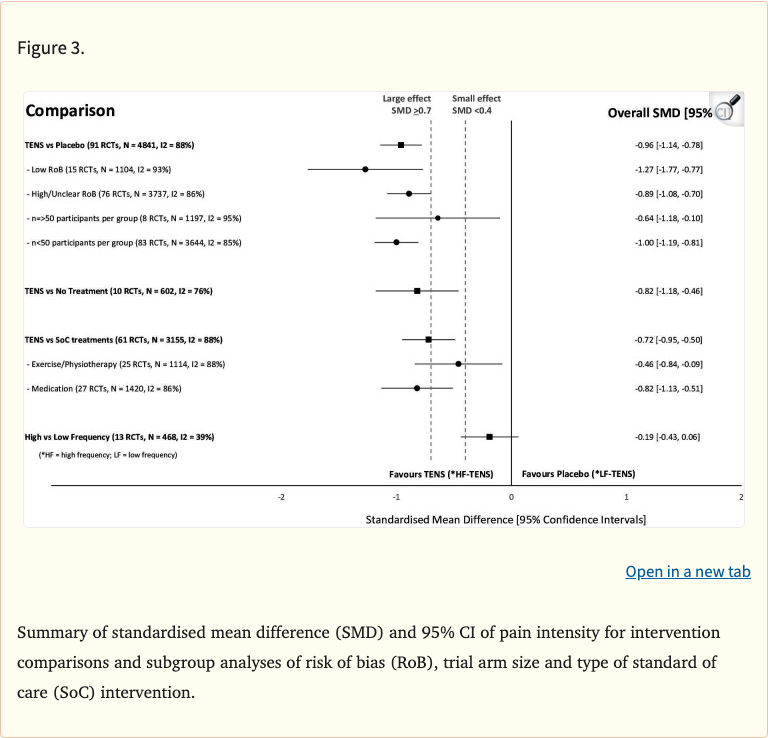 Figure 1
Figure 1
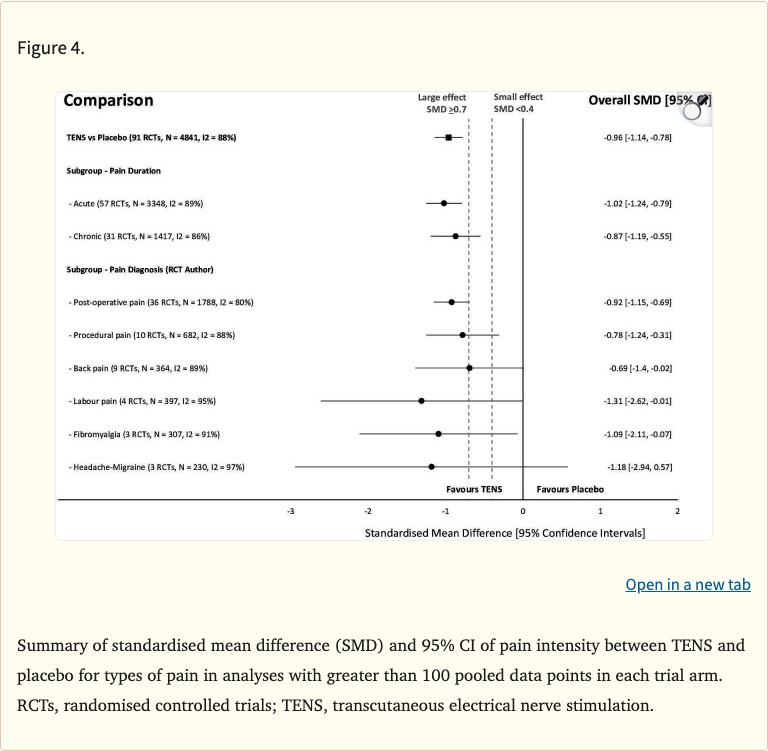 Figure 2
Figure 2
Limitations
- High heterogeneity across studies due to varying TENS parameters
- Many studies had methodological limitations (blinding, allocation concealment)
- Publication bias possible despite comprehensive search
- Optimal TENS parameters could not be definitively determined
- Long-term follow-up data limited
- Some evidence downgraded due to risk of bias
- Cost-effectiveness not assessed