Key Takeaway
C15:0 showed broader and safer cell-based activities than omega-3 EPA across 12 human disease systems
Summary
Study comparing the cell-based activities of pentadecanoic acid (C15:0) to eicosapentaenoic acid (EPA/omega-3) across multiple human disease systems.
Methods
- Tested C15:0 and EPA in 12 primary human cell-based disease systems
- Dose-dependent evaluations at physiologically relevant concentrations
- Assessed activities across cardiometabolic, immune, and cancer systems
- Compared safety profiles between fatty acids
Key Results
- C15:0 had clinically relevant activities across more systems than EPA
- C15:0 showed safer profile with fewer off-target effects
- Activities paralleled common therapeutics for mood, infection, cancer
- Supports C15:0 as emerging essential fatty acid
Figures
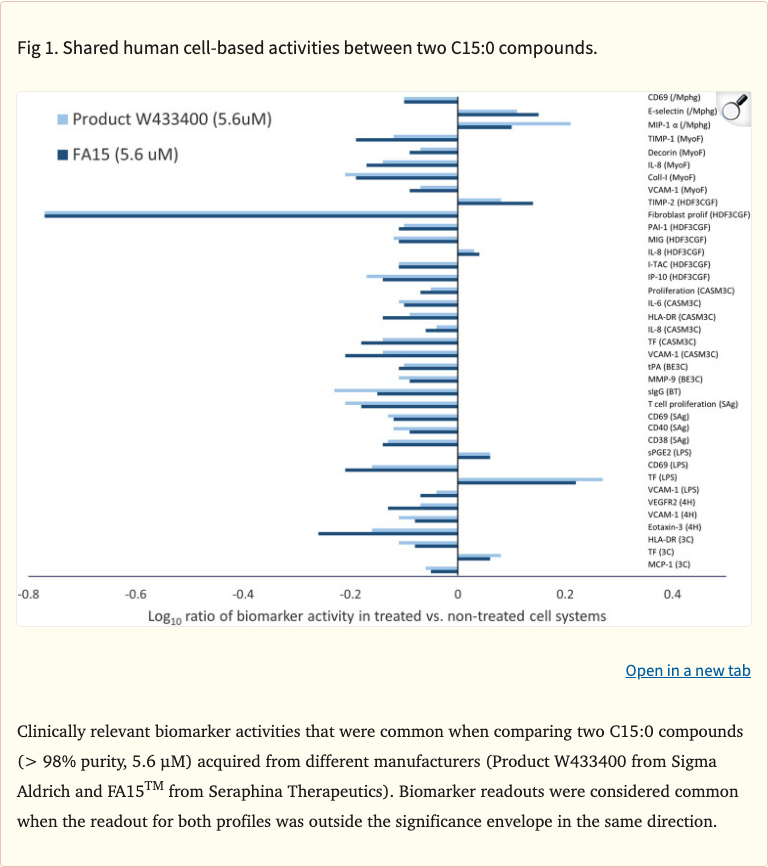 Figure 1
Figure 1
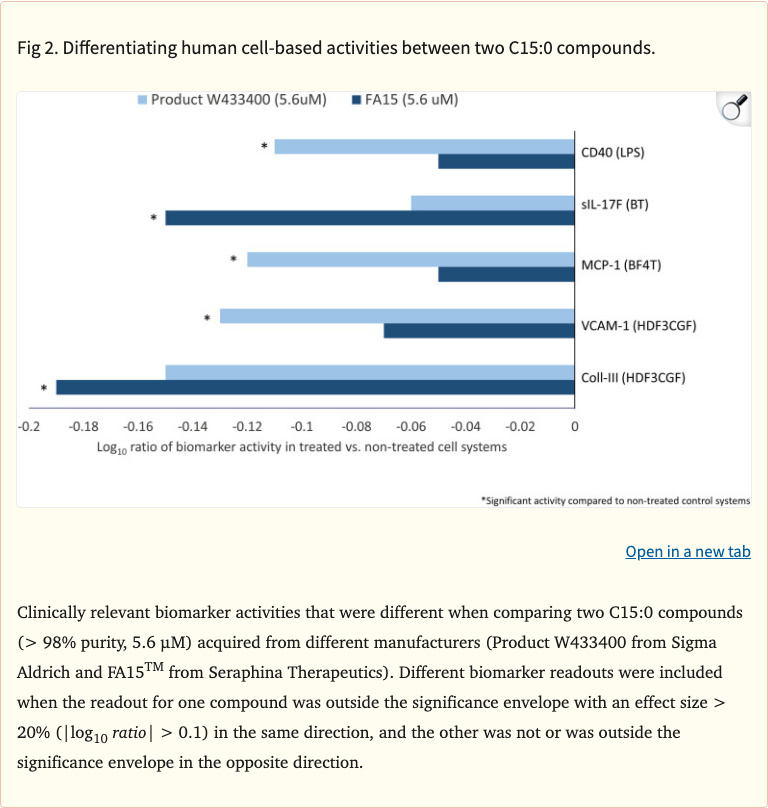 Figure 2
Figure 2
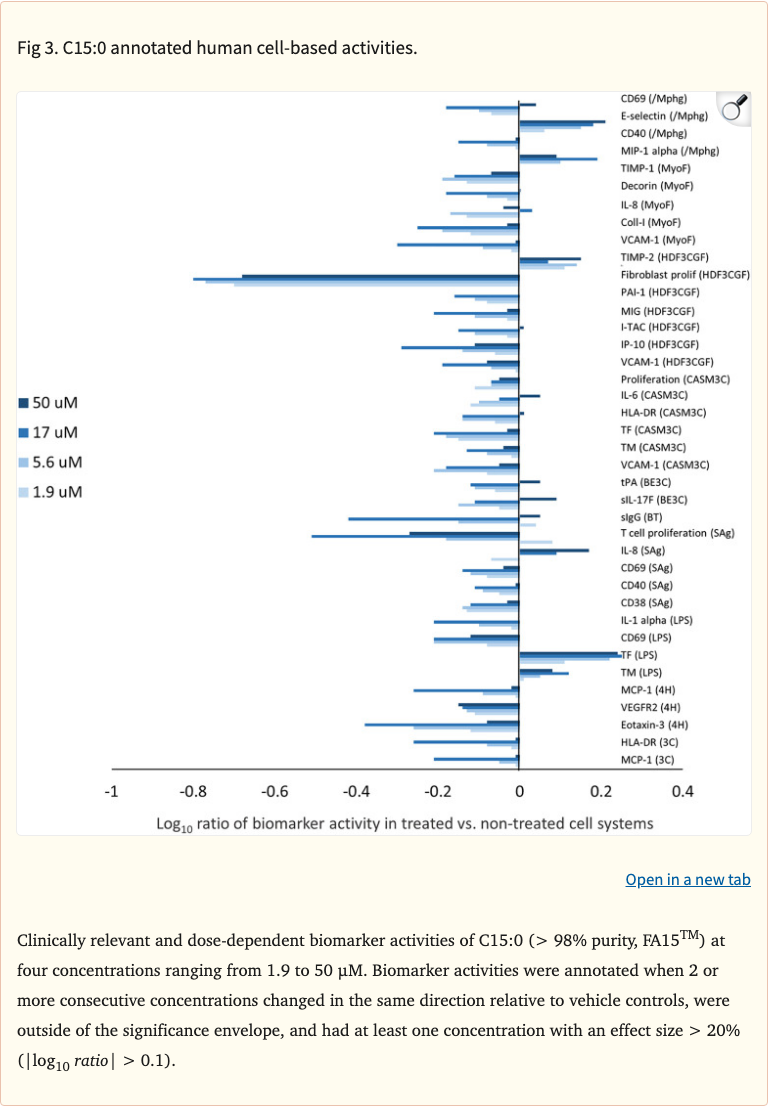 Figure 3
Figure 3
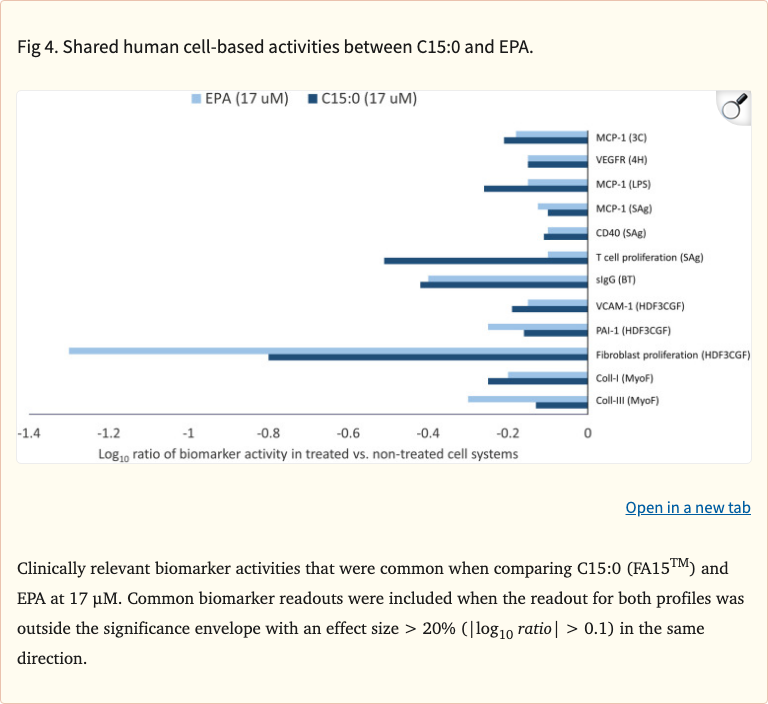 Figure 4
Figure 4
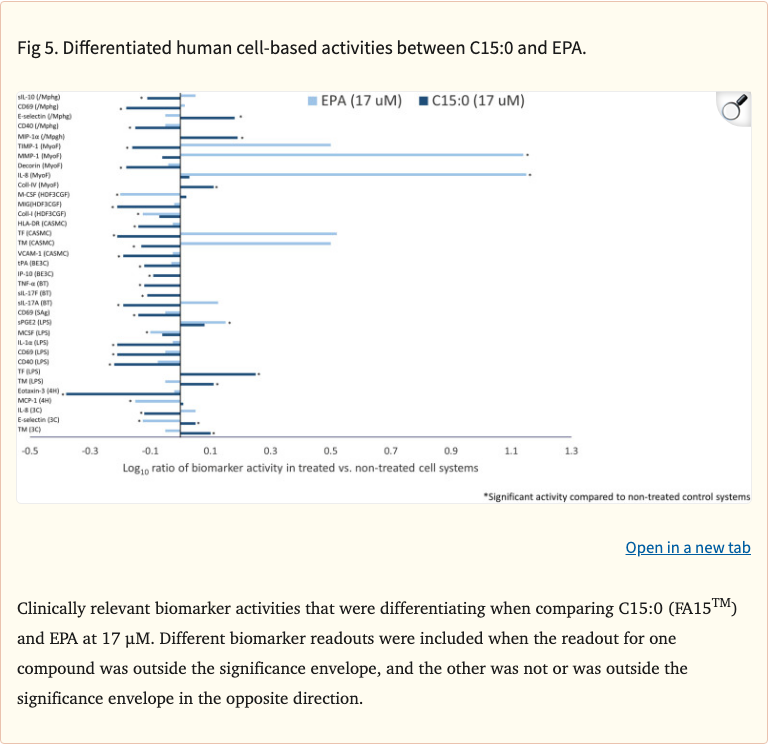 Figure 5
Figure 5
Limitations
- Cell-based study (not human clinical trial)
- In vitro results may not translate to in vivo
- Authors have commercial interest in C15:0 supplements
- Limited long-term safety data