Key Takeaway
Across 29 RCTs with 3,846 participants, 65.5% of studies found silymarin reduced liver enzyme levels, supporting its hepatoprotective potential especially in NAFLD patients.
Summary
This systematic review followed PRISMA 2020 guidelines to examine randomized clinical trials published between 1992 and 2023 evaluating silymarin's effects on liver enzymes. The authors searched PubMed/MEDLINE and analyzed 29 RCTs involving 3,846 participants with various underlying liver conditions.
The review found that 65.5% of studies reported reduced liver enzyme levels with silymarin supplementation, 20.7% showed no significant change, and 13.8% observed elevated liver enzymes. Dosages across studies ranged from 140 to 420 mg daily with variable treatment durations.
The authors concluded that silymarin demonstrates a potential advantageous influence on liver enzyme levels, indicating hepatoprotective potential. Effects appeared context-dependent, with particularly positive outcomes in non-alcoholic fatty liver disease patients. They noted that outcome disparities may stem from comorbidities, suboptimal doses, and underlying diseases.
Methods
Systematic review following PRISMA 2020 guidelines. Searched PubMed/MEDLINE for randomized clinical trials from 1992-2023 examining silymarin supplementation and liver enzyme outcomes. 29 RCTs with 3,846 total participants were included. Risk of bias assessed using the Cochrane Risk of Bias 2.0 tool.
Key Results
- 65.5% of studies (19/29) reported reduced liver enzyme levels
- 20.7% of studies (6/29) showed no significant change
- 13.8% of studies (4/29) observed elevated liver enzymes
- Silymarin dosages ranged from 140-420 mg daily
- Strongest benefits seen in NAFLD patient populations
Figures
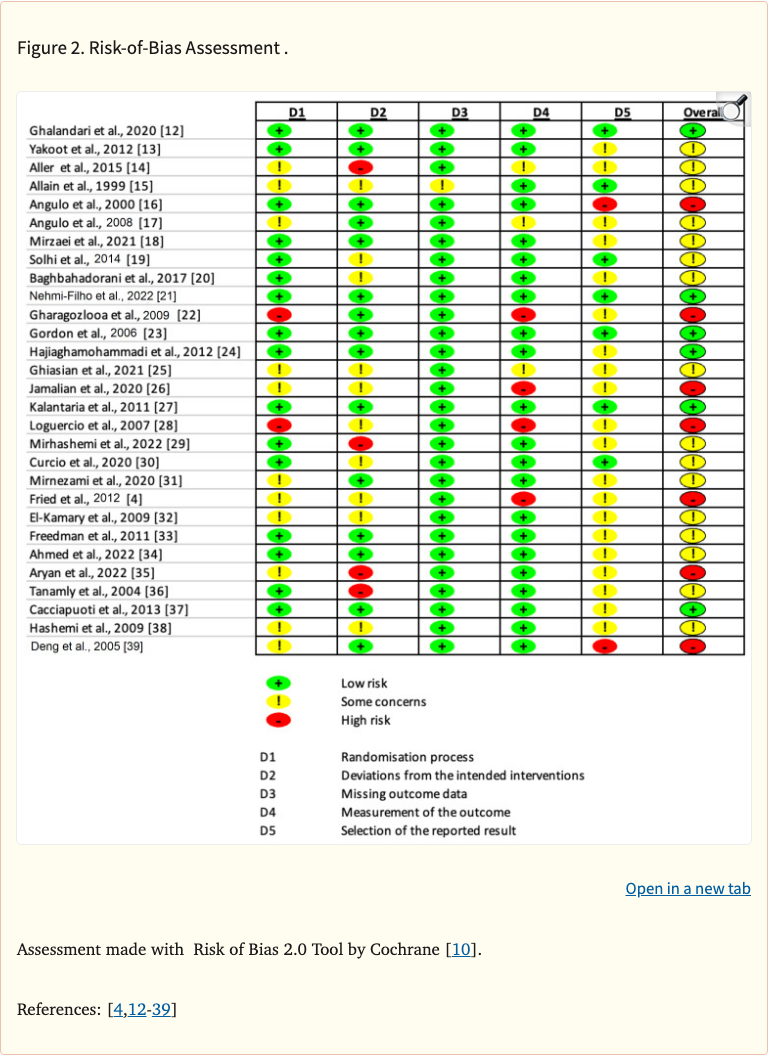 Figure 1
Figure 1
Limitations
- High heterogeneity across included studies in patient populations and dosing
- Variable treatment durations make dose-response assessment difficult
- Searched only PubMed/MEDLINE, possibly missing relevant studies from other databases
- Outcome disparities may stem from comorbidities and suboptimal dosing
- No formal meta-analytic pooling of effect sizes reported
