Key Takeaway
Prolonged water fasting (2-21+ days) produces significant weight loss and metabolic improvements, but evidence quality is limited and refeeding requires careful management.
Summary
This narrative review synthesized findings from human trials on prolonged water fasting (PWF), defined as water-only fasting lasting 2 or more consecutive days. The authors examined studies ranging from short-term clinical trials to observational data from fasting clinics like Buchinger Wilhelmi.
Key findings across studies included substantial weight loss (typically 0.5-1 kg per day, mostly water and glycogen initially), reductions in blood pressure, improvements in lipid profiles, and decreased fasting glucose and insulin levels. The review also examined autophagy activation and the metabolic shift to ketosis that occurs during extended fasts.
However, the authors noted significant limitations in the existing evidence base. Most studies lacked control groups, had small sample sizes, and follow-up was typically short. The review emphasized that proper medical supervision and careful refeeding protocols are essential, particularly for fasts exceeding 5-7 days, due to refeeding syndrome risk.
Methods
- Narrative review methodology (not systematic)
- Searched PubMed and other databases for human trials on prolonged water fasting
- Included studies with fasting durations of 2+ consecutive days
- Examined both controlled trials and observational studies from fasting clinics
- Focused on metabolic, cardiovascular, and body composition outcomes
Key Results
- Weight loss: 0.5-1 kg/day during fasting, with significant water and glycogen losses initially
- Blood pressure: Consistent reductions observed across studies, some dramatic
- Lipids: Improvements in total cholesterol, LDL, and triglycerides in most studies
- Glucose/insulin: Fasting glucose and insulin levels decreased during fasts
- Autophagy markers: Limited human data, but animal studies suggest upregulation by 24-48 hours
- Safety: Generally well-tolerated with medical supervision; minor side effects common (headache, fatigue)
- Refeeding: Critical phase requiring gradual reintroduction of food to avoid refeeding syndrome
Figures
 Figure 1
Figure 1
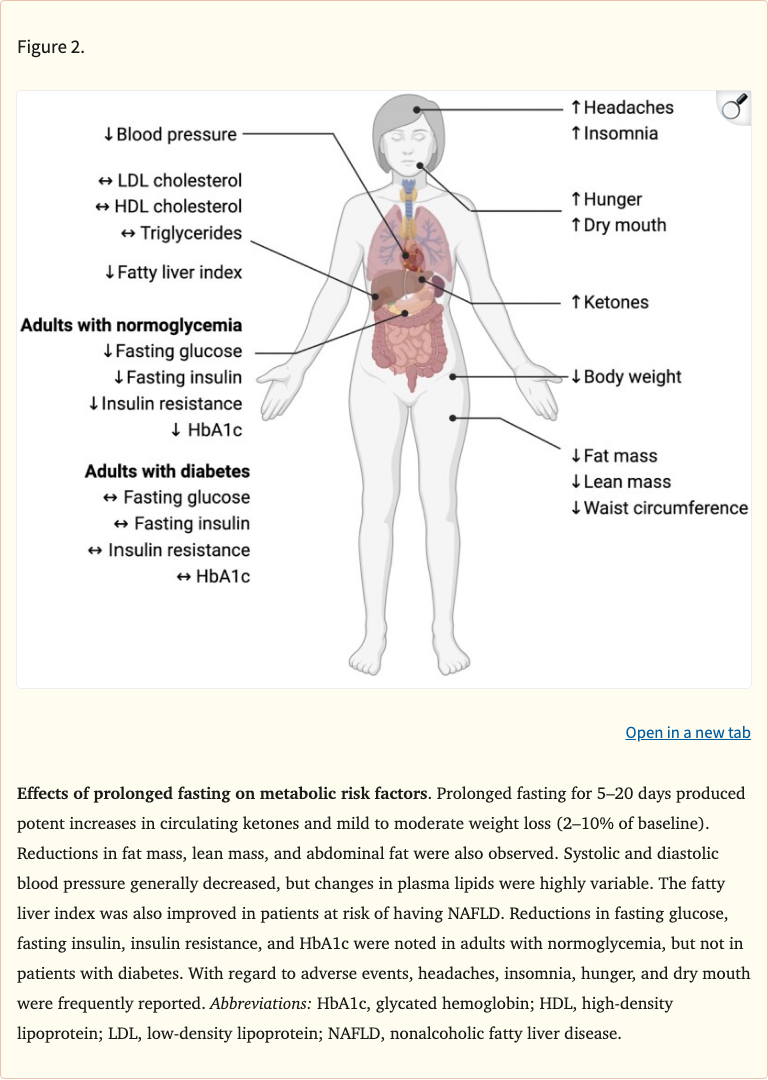 Figure 2
Figure 2
Limitations
- Narrative review (not systematic), potential selection bias
- Most included studies lacked control groups
- Small sample sizes in clinical trials
- Short follow-up periods - unclear if benefits persist
- Heterogeneous fasting protocols and durations
- Many studies from fasting clinics with self-selected populations
- Limited data on long-term safety of repeated extended fasts
- Autophagy measurements in humans remain challenging
