Key Takeaway
PEMF therapy reduced pain by 36% vs 10% for standard care and cut medication use by 55% in a multi-center RCT of 91 patients with joint and soft tissue injuries.
Summary
This prospective, multi-center randomized clinical trial evaluated pulsed electromagnetic field (PEMF) therapy for managing joint and soft tissue pain across five orthopedic clinics. A total of 120 patients were enrolled, with complete data from 91 participants (48 PEMF, 43 standard-of-care). The PEMF group self-administered daily therapy using the Orthocor Active System, while the control group received clinician-prescribed standard treatment.
After 14 days, the PEMF group showed a 36% reduction in pain scores (least squares mean change of -1.8), compared to only 10% reduction (-0.46) in the standard-of-care group, a highly significant difference (p < 0.0001). Medication use in the PEMF group dropped from 40% to 18% (a 55% reduction), versus only a modest decrease from 40% to 35% in controls.
A crossover phase allowed standard-of-care patients to switch to PEMF for an additional 16 days. These crossover patients experienced an additional 18% decrease in pain scores and 63% decrease in pharmacologic use, further supporting PEMF's efficacy. The authors concluded that PEMF was significantly more effective than standard care for pain management and recommended it as a noninvasive treatment option.
Methods
- Prospective, multi-center, randomized clinical trial
- 5 orthopedic clinic sites
- 120 patients enrolled, 91 with complete data (48 PEMF, 43 SOC)
- Joint and soft tissue pain patients
- PEMF group: self-administered daily Orthocor Active System
- SOC group: clinician-prescribed standard treatment
- Initial treatment phase: 14 days
- Crossover phase: SOC patients could switch to PEMF for 16 additional days
- Primary outcomes: pain score change and medication use
Key Results
- PEMF group: 36% pain reduction (LS mean change -1.8 from baseline)
- SOC group: 10% pain reduction (LS mean change -0.46)
- Difference highly significant (p < 0.0001)
- PEMF medication use dropped from 40% to 18% (55% reduction)
- SOC medication use dropped from 40% to 35% (12% reduction)
- Crossover patients: additional 18% pain decrease after switching to PEMF
- Crossover patients: 63% decrease in pharmacologic use
Figures
 Figure 1
Figure 1
 Figure 2
Figure 2
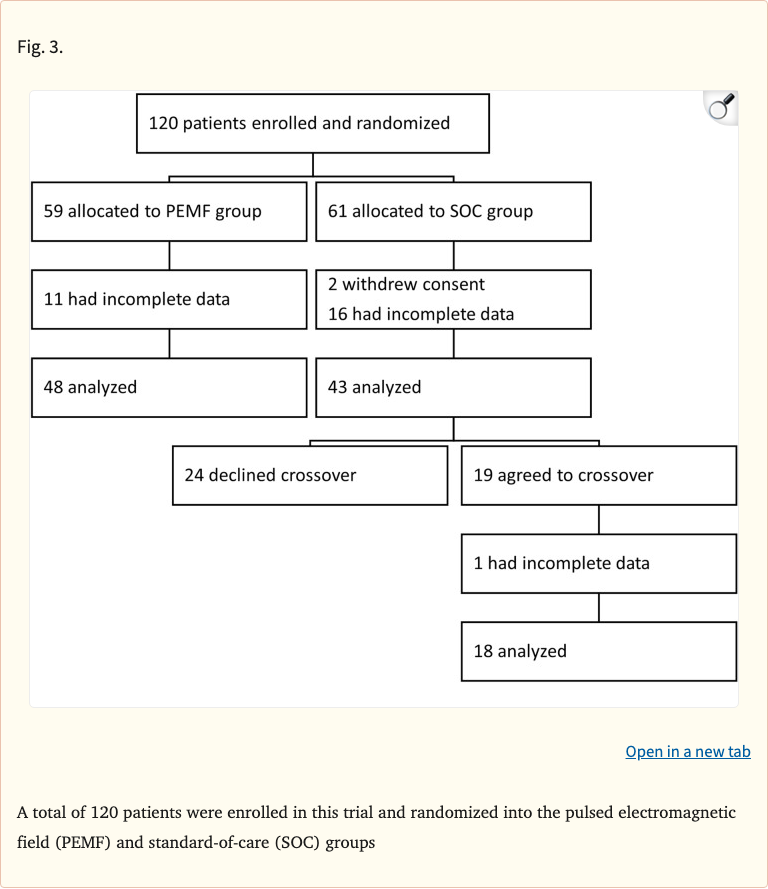 Figure 3
Figure 3
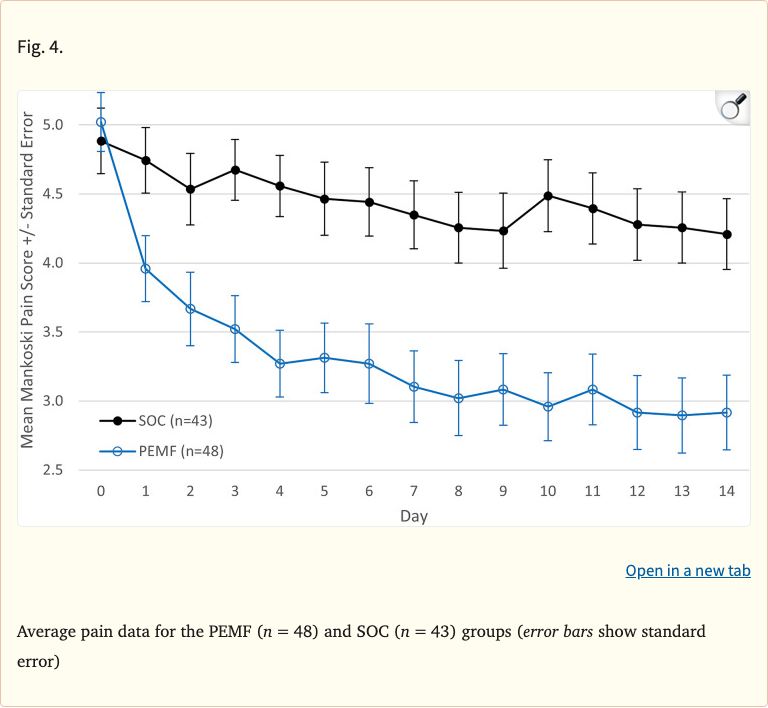 Figure 4
Figure 4
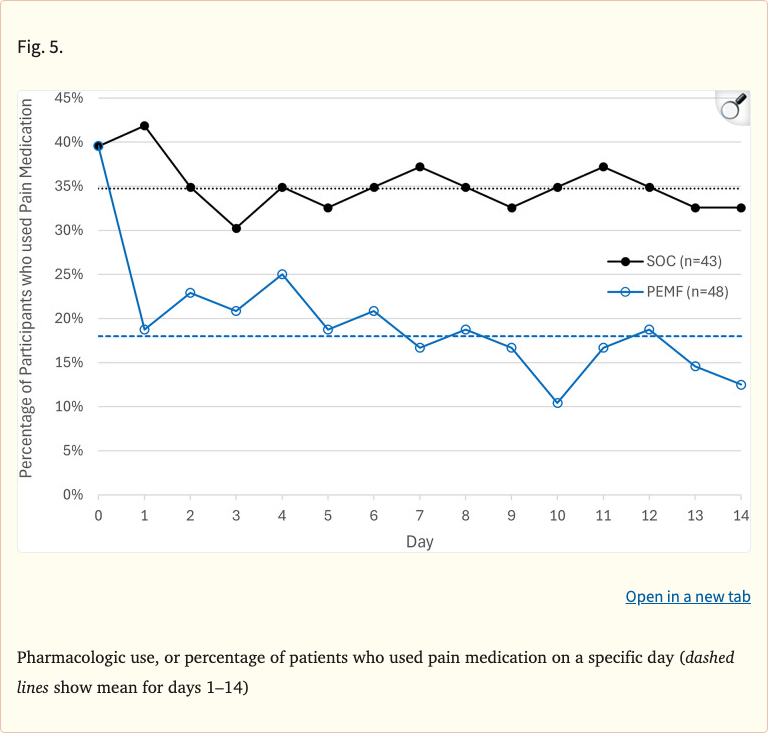 Figure 5
Figure 5
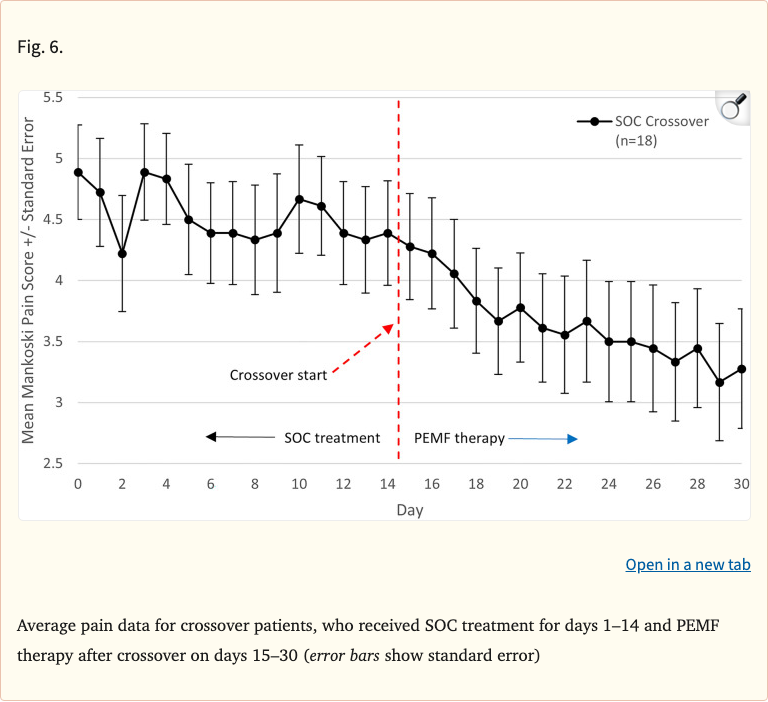 Figure 6
Figure 6
Limitations
- Moderate sample size (91 completers)
- Short treatment duration (14 days initial phase)
- Single PEMF device (Orthocor Active System) tested
- Not blinded (open-label design)
- Limited long-term follow-up
- Industry involvement possible (specific device used)
