Key Takeaway
In healthy adults 70+, daily low-dose aspirin did not prolong disability-free survival and increased major bleeding risk.
Summary
ASPREE was a landmark randomized, double-blind, placebo-controlled trial examining whether daily low-dose aspirin (100mg) would extend healthy, independent life in older adults.
Study Design:
- 19,114 community-dwelling adults aged 70+ (65+ for US minorities)
- Median follow-up: 4.7 years
- Primary endpoint: Disability-free survival (survival free of dementia or persistent physical disability)
Key Results:
No Benefit for Primary Endpoint:
- Disability-free survival: No significant difference between aspirin and placebo
- Aspirin group: 21.5 events per 1000 person-years
- Placebo group: 21.2 events per 1000 person-years
Increased Bleeding:
- Major hemorrhage: 3.8% aspirin vs 2.8% placebo (HR 1.38)
- Upper GI bleeding was the most common site
- Intracranial bleeding also increased
Unexpected Cancer Signal:
- Higher cancer-related mortality in aspirin group (3.1% vs 2.3%)
- This finding was unexpected and requires further investigation
Conclusions:
This trial was pivotal in changing guidelines to recommend against initiating aspirin for primary prevention in adults 70 and older. The bleeding risks clearly outweighed any potential benefits in this population.
Figures
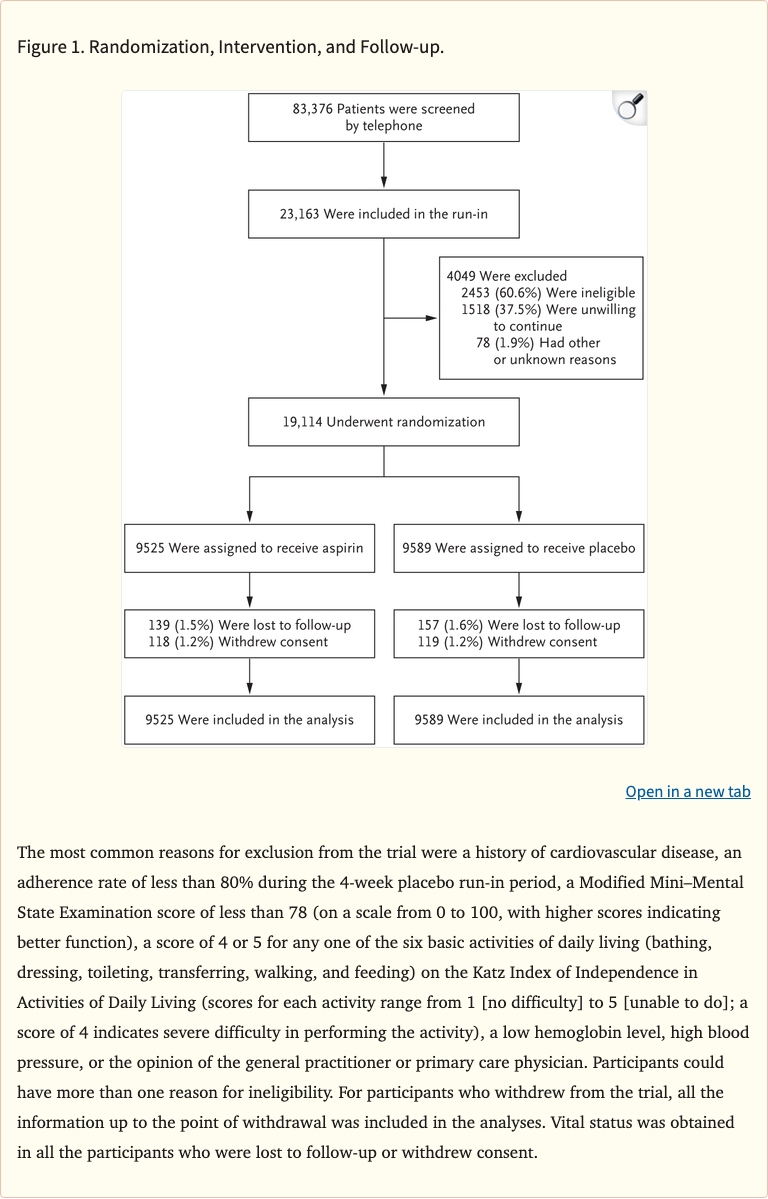 Figure 1
Figure 1
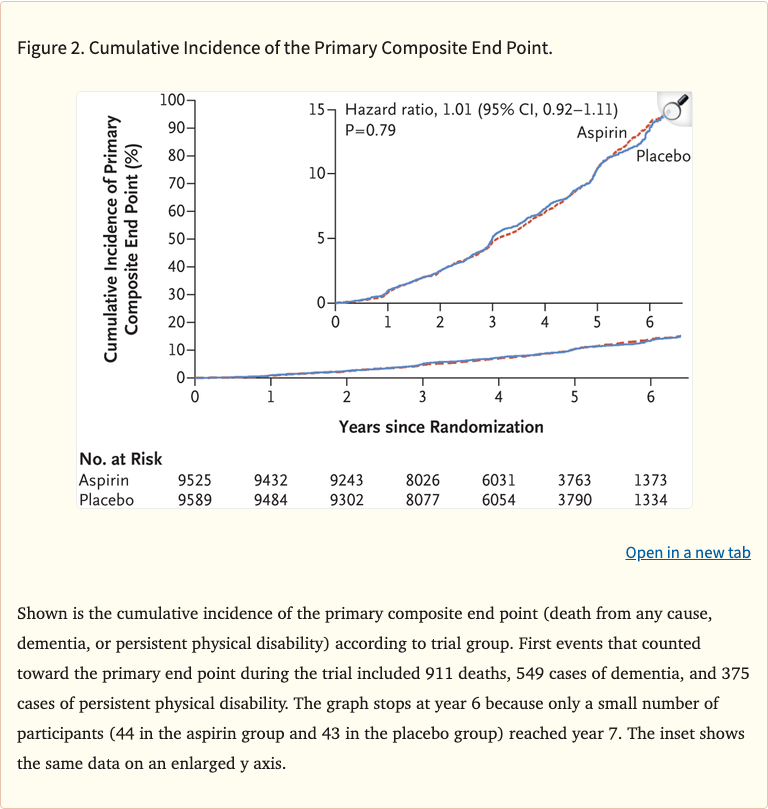 Figure 2
Figure 2
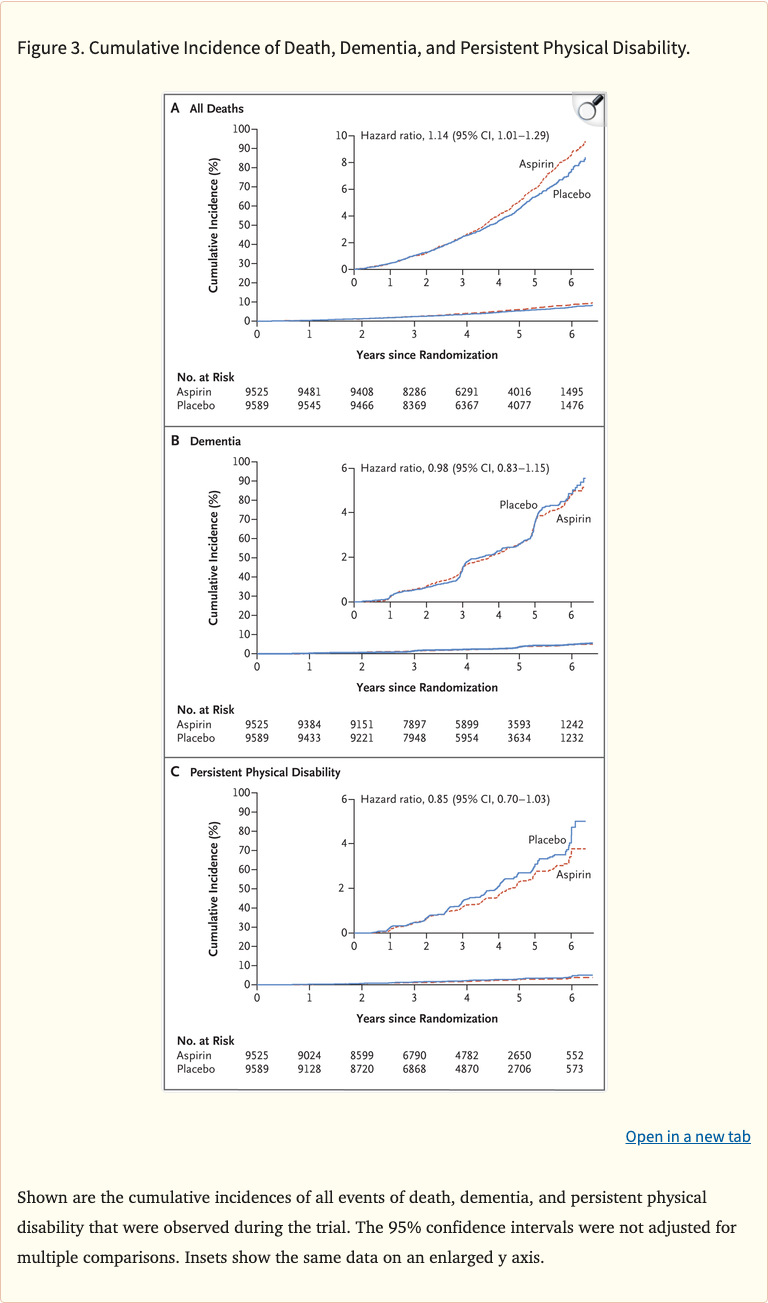 Figure 3
Figure 3
