Key Takeaway
PMR significantly improved sleep quality (SMD -1.43) and reduced anxiety (SMD -2.56) in COVID-19 patients across 9 studies with 626 participants.
Summary
This systematic review and meta-analysis evaluated the effects of progressive muscle relaxation (PMR) exercises on sleep quality, anxiety, and depression in patients with COVID-19. The authors searched major databases including PubMed, Cochrane Library, Web of Science, and Scopus for randomized controlled trials examining PMR in COVID-19 patients.
Nine RCTs with a total of 626 participants were included. The meta-analysis found that PMR significantly improved sleep quality (SMD -1.43, 95% CI: -2.37 to -0.49) and reduced anxiety levels (SMD -2.56, 95% CI: -4.21 to -0.91) compared to control groups. However, the effect on depression did not reach statistical significance.
The findings suggest that PMR is a safe, accessible, and effective non-pharmacological intervention for managing the psychological burden of COVID-19, particularly for sleep disturbances and anxiety. The authors recommend integrating PMR into standard care protocols for hospitalized COVID-19 patients.
Methods
Systematic review and meta-analysis of RCTs following PRISMA guidelines. Searched PubMed, Cochrane Library, Web of Science, and Scopus. Included 9 RCTs with 626 COVID-19 patients. Quality assessed using the Cochrane risk of bias tool. Effect sizes calculated as standardized mean differences (SMD) with 95% confidence intervals using random-effects models.
Key Results
PMR significantly improved sleep quality (SMD -1.43, 95% CI: -2.37 to -0.49, p = 0.003) and reduced anxiety (SMD -2.56, 95% CI: -4.21 to -0.91, p = 0.002). The effect on depression was not statistically significant. Substantial heterogeneity was observed across studies (I-squared > 75% for most outcomes).
Figures
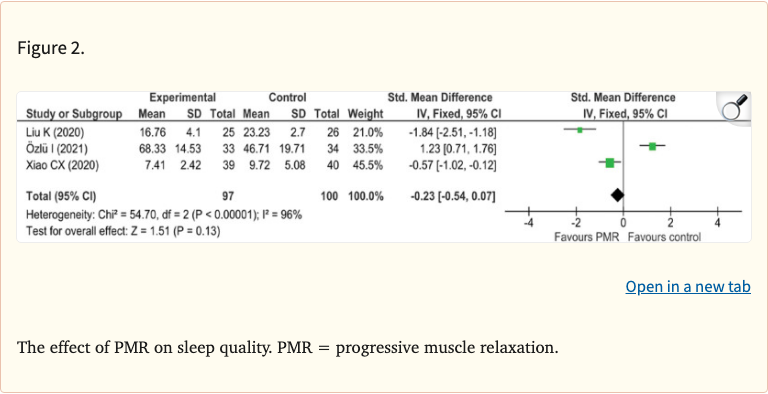 Figure 1
Figure 1
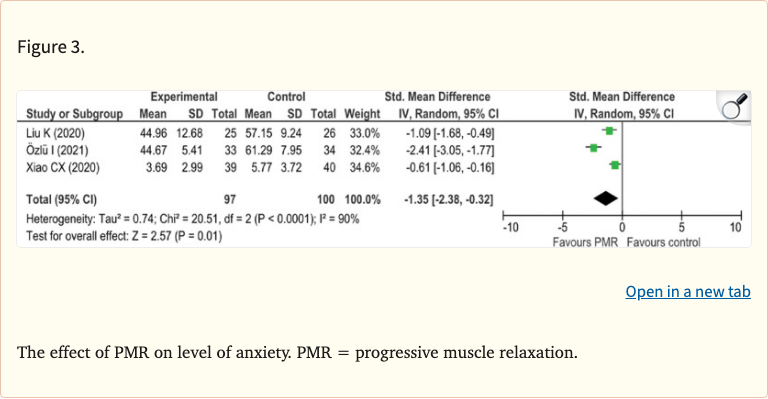 Figure 2
Figure 2
Limitations
High heterogeneity across included studies. Relatively small total sample size (626 participants). Most studies were conducted in China, limiting generalizability. Variation in PMR protocols (duration, frequency, follow-up periods) across studies. Risk of bias concerns in some included trials due to lack of blinding.
