Key Takeaway
In 72 adults with anxiety and poor sleep, CBD (25-175mg/day) reduced anxiety scores in 79.2% and improved sleep in 66.7% within the first month
Summary
This large retrospective case series from a psychiatric clinic examined CBD as an adjunct to standard treatment for anxiety and sleep complaints. Results showed rapid improvements in both anxiety and sleep scores with good tolerability, providing real-world evidence for CBD's therapeutic potential.
Methods
- Design: Retrospective case series
- Setting: Psychiatric outpatient clinic
- Participants: 72 adults with primary anxiety or sleep concerns
- Intervention: CBD 25-175mg/day as adjunct to standard care
- Duration: Monthly assessments over 3+ months
- Measures: Hamilton Anxiety Rating Scale (HAM-A), Pittsburgh Sleep Quality Index (PSQI)
Key Results
- Anxiety outcomes:
- 79.2% (57/72) showed decreased anxiety in first month
- Anxiety scores remained lower throughout study period
- Average HAM-A scores decreased significantly
- Sleep outcomes:
- 66.7% (48/72) showed improved sleep in first month
- Sleep improvements fluctuated more over time
- More variable response than anxiety
- Dosing:
- Most patients received 25mg/day in capsule form
- Some increased to 50-75mg/day
- Doses ranged from 25-175mg/day
- Tolerability:
- CBD was well-tolerated overall
- 3 patients reported mild side effects
- No patients discontinued due to side effects
Figures
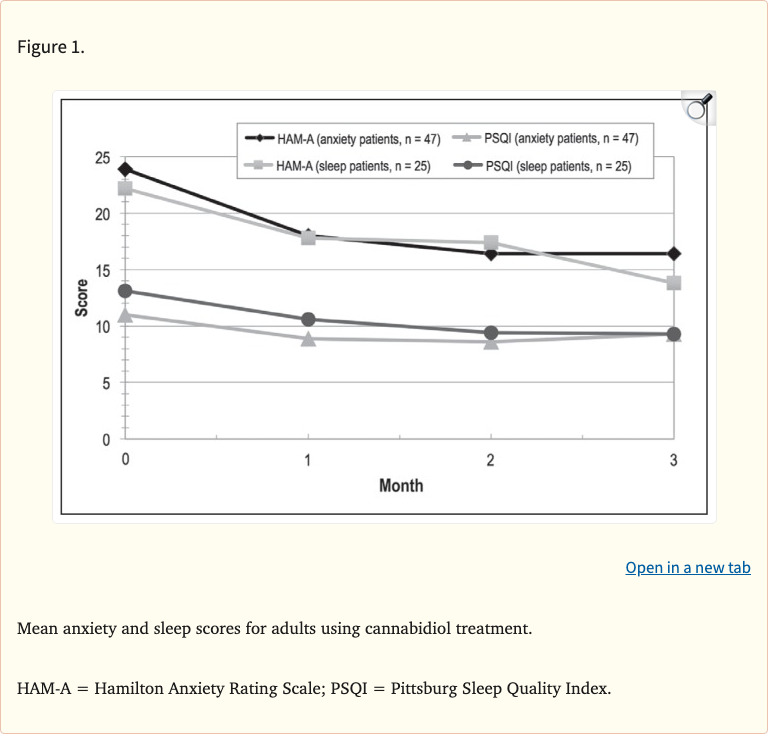 Figure 1
Figure 1
Limitations
- Retrospective, uncontrolled design
- No placebo group - cannot rule out placebo effect
- Variable dosing across patients
- CBD used as adjunct, not monotherapy
- Short follow-up for some patients
- Selection bias possible in clinical population