Key Takeaway
Melatonin appears safe for short-term use in children with no increase in serious adverse events, though long-term safety data remains limited.
Summary
This systematic review comprehensively assessed the safety profile of melatonin in children and adolescents, examining both short-term and long-term adverse effects from randomized controlled trials and observational studies.
The review found that melatonin was well-tolerated in the short term, with no significant increase in serious adverse events compared to placebo. Common mild side effects included morning drowsiness, headache, and dizziness, but these were generally comparable to placebo groups. The evidence for long-term safety was limited due to the short duration of most available studies.
Using GRADE methodology, the authors assessed the certainty of evidence for various adverse outcomes. They concluded that while short-term use appears safe, clinicians should remain cautious about prolonged use in children given the lack of robust long-term data, particularly regarding effects on pubertal development.
Methods
- Systematic review of RCTs and observational studies
- Searched PubMed, Embase, Cochrane Library, and clinical trial registries
- Included studies reporting adverse events in children/adolescents taking melatonin
- Categorized adverse events by type and timing (short-term vs long-term)
- GRADE assessment for each adverse outcome category
- Narrative synthesis for long-term observational data
Key Results
- No increase in serious adverse events vs placebo in RCTs
- Common mild effects: drowsiness, headache, dizziness (similar to placebo rates)
- No significant impact on growth or pubertal development in short-term studies
- Limited long-term data available (most studies <6 months)
- Low to very low certainty evidence for most outcomes
Figures
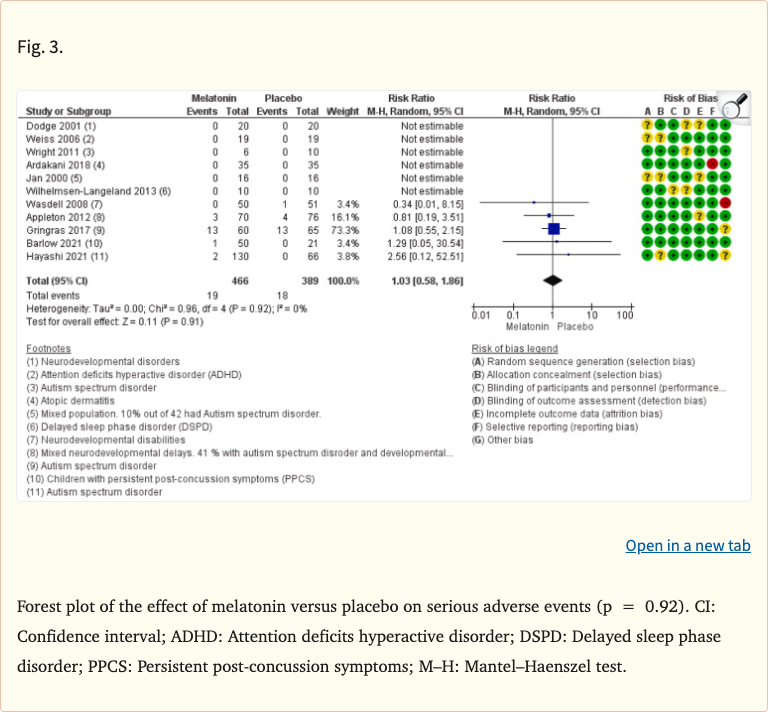 Figure 1
Figure 1
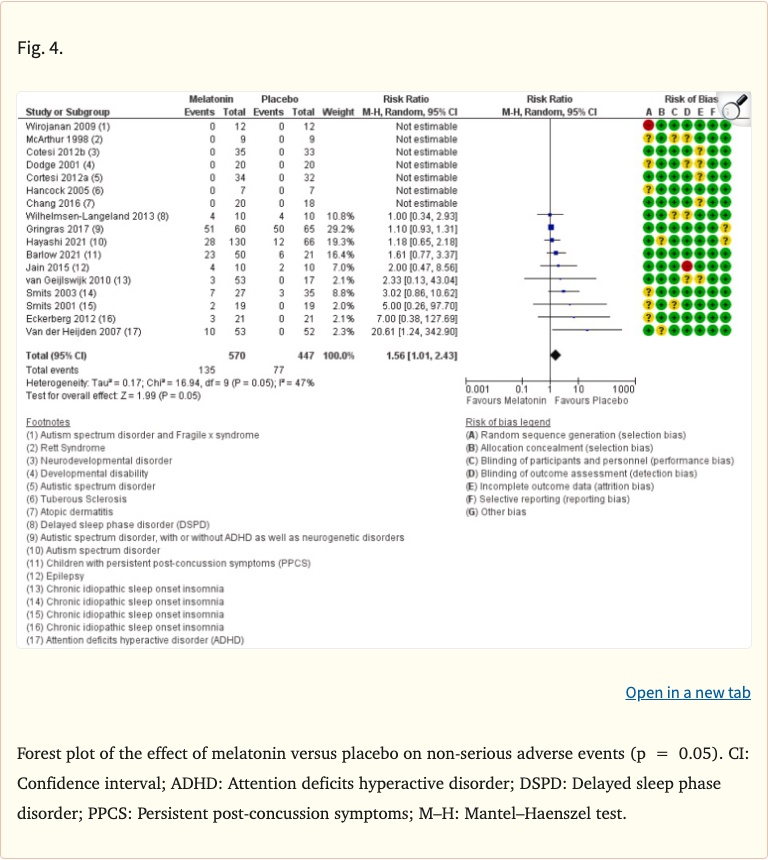 Figure 2
Figure 2
Limitations
- Most RCTs had short follow-up periods (weeks to months)
- Inconsistent adverse event reporting across studies
- Limited data on doses >5mg
- Observational long-term data prone to confounding
- Pubertal development outcomes rarely assessed
