Key Takeaway
A dose-dependent RCT found that 300, 600, and 900 mg/day NMN safely increased blood NAD+ levels and improved walking endurance in healthy middle-aged adults over 60 days.
Summary
This rigorous multicenter, double-blind, placebo-controlled trial investigated the dose-dependent effects of NMN supplementation in healthy middle-aged adults aged 40-65. Participants were randomized to receive 300 mg, 600 mg, or 900 mg of NMN per day, or placebo, for 60 days. The study measured blood NAD+ concentrations, clinical safety markers, and physical performance outcomes including the six-minute walk test.
All three NMN doses significantly increased blood NAD+ levels compared to placebo, confirming oral NMN bioavailability in humans. The 600 mg and 900 mg groups showed the most pronounced increases. Notably, the study demonstrated improvements in walking endurance as measured by the six-minute walk test, suggesting functional physical benefits from NMN supplementation.
Safety assessments showed NMN was well-tolerated at all doses with no serious adverse events. Clinical laboratory values, vital signs, and adverse event profiles were comparable between NMN and placebo groups. This study is particularly valuable as one of the first well-designed dose-response trials in humans, providing evidence for both the safety and efficacy of NMN at commonly used supplement doses.
Methods
Randomized, multicenter, double-blind, placebo-controlled, parallel-group, dose-dependent clinical trial. 80 healthy middle-aged adults (40-65 years) were randomized to four groups: placebo, 300 mg/day NMN, 600 mg/day NMN, or 900 mg/day NMN for 60 days. Primary outcomes included blood NAD+ concentrations and safety markers. Secondary outcomes included the six-minute walk test, SF-36 quality of life scores, and clinical laboratory assessments. Blood samples were collected at baseline, day 30, and day 60.
Key Results
- All three NMN doses significantly increased blood NAD+ levels compared to placebo (p < 0.05)
- 600 mg and 900 mg groups showed the greatest NAD+ increases
- Six-minute walk test distance improved significantly in NMN groups vs placebo
- SF-36 quality of life scores showed trends toward improvement
- No dose-limiting toxicities or serious adverse events at any dose level
- Clinical laboratory values remained within normal ranges across all groups
- NAD+ increase was observable by day 30 and maintained through day 60
Figures
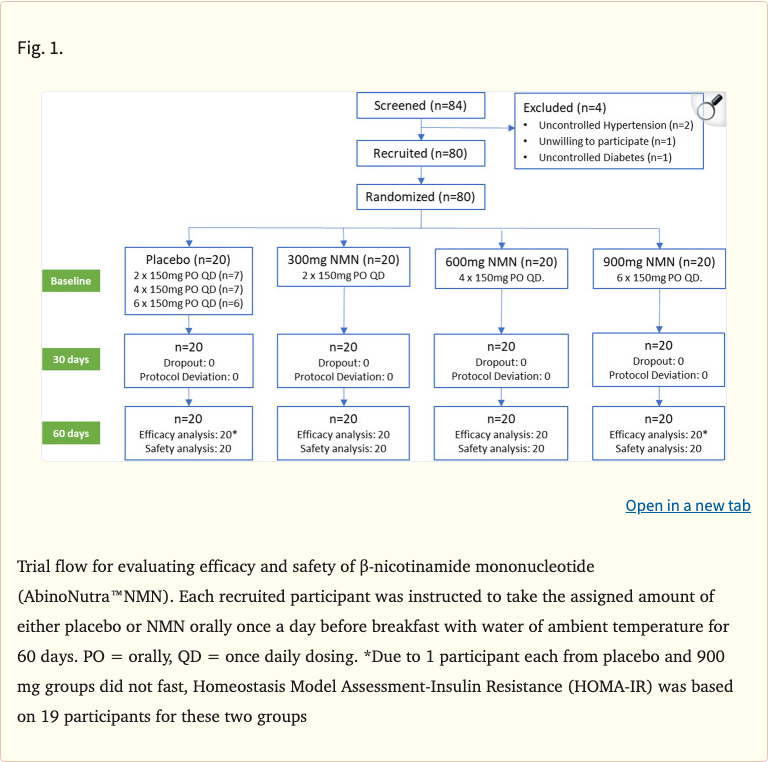 Figure 1
Figure 1
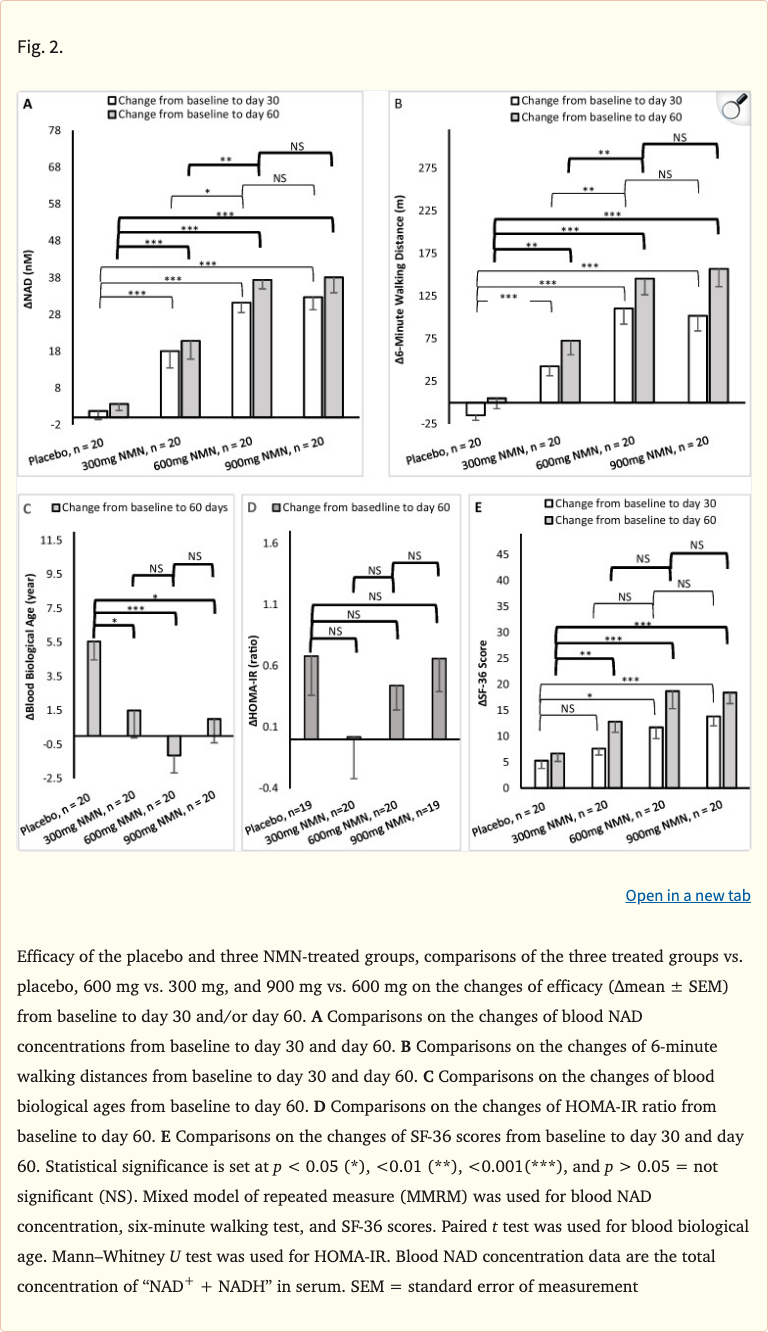 Figure 2
Figure 2
Limitations
- Relatively small sample size (80 participants total, ~20 per group)
- Short duration (60 days) - long-term effects not assessed
- Conducted in healthy adults only - effects in those with metabolic conditions may differ
- Single ethnicity population limits generalizability
- Industry-funded study (Uthever NMN manufacturer)
- Did not measure intracellular NAD+ or tissue-specific levels
- Walking endurance may not reflect broader health outcomes
