Key Takeaway
Across 177 studies (6,322 subjects), taVNS showed no difference in adverse event risk vs sham, with only mild transient effects like ear pain and tingling.
Summary
This large systematic review and meta-analysis evaluated the safety profile of transcutaneous auricular vagus nerve stimulation (taVNS) across 177 studies encompassing 6,322 subjects. The authors searched Medline, Embase, Web of Science, Cochrane, and Lilacs databases through April 2022 to comprehensively assess adverse event reporting and risk.
A notable finding was the poor state of safety reporting: 55% of studies did not mention the presence or absence of any adverse events at all, and only about 25% described at least one adverse event. Among studies that did report adverse events, the incidence rate was 12.84 per 100,000 person-minutes-days of stimulation. The most commonly reported effects were ear pain, headache, and tingling at the stimulation site - all mild and transient.
Critically, the meta-analysis found no statistically significant difference in the risk of developing an adverse event between active taVNS and sham/control conditions. No causal relationship was identified between taVNS and any severe adverse events. The authors concluded that taVNS is a safe and feasible option for clinical intervention, though they emphasized the need for improved adverse event reporting standards in future research.
Methods
- Systematic search of 5 databases (Medline, Embase, Web of Science, Cochrane, Lilacs) through April 7, 2022
- 177 studies with 6,322 total subjects included
- Assessed adverse event reporting practices across all included studies
- Meta-analytic comparison of adverse event risk between active taVNS and sham/control groups
- Calculated adverse event incidence rate per person-minutes-days of stimulation
Key Results
- 55.37% of studies did not mention presence or absence of any adverse events
- Only 24.86% of studies described at least one adverse event
- Adverse event incidence rate: 12.84 per 100,000 person-minutes-days of stimulation
- Most common adverse events: ear pain, headache, and tingling
- No significant difference in adverse event risk between active taVNS and controls
- No causal relationship identified between taVNS and severe adverse events
- All reported adverse events were mild and transient
Figures
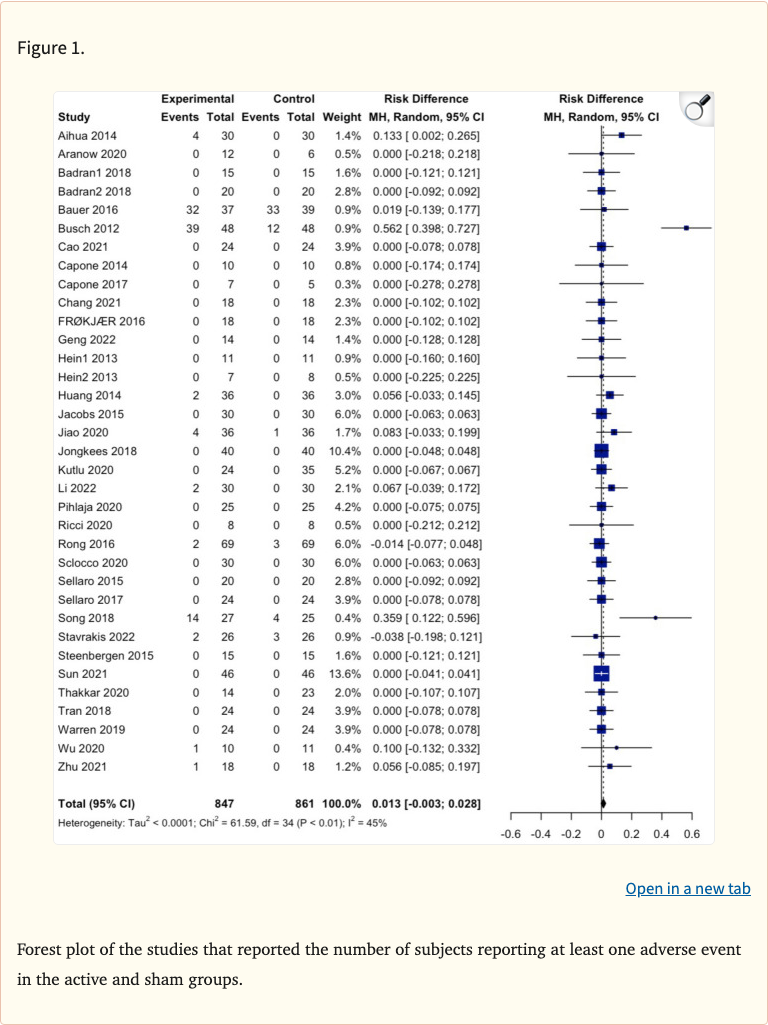 Figure 1
Figure 1
Limitations
- Over half of included studies did not report on adverse events at all
- Heterogeneity in stimulation parameters across studies
- Inconsistent definitions and reporting of adverse events
- Limited long-term safety data
- Publication bias may underreport adverse events
